Neck Pain Solutions With Quality Products Used At Home
 Seeking neck pain solutions, we can fortunately rely on technology and innovation to produce effective neck pain relief products that can help.
Seeking neck pain solutions, we can fortunately rely on technology and innovation to produce effective neck pain relief products that can help.
Some types of neck pain will require invasive measures like spinal injections or post-surgical relief, however, most neck pain begins with issues related to posture and minor strains, which progress over time to more serious issues.
Cervical spine problems are on the rise. With the advent of technology comes long hours or working at computers and using laptops, tablets and cellphones. Even much of leisure time is spent using these devices and it has negative effects on the health of the cervical spine, like the commonly seen forward head posture. With the addition of accidents, injuries and other tasks which require awkward neck postures, more effective means are required to combat a growing epidemic.
Pain is a sign that something is wrong. If proper attention is given at the first signs of pain, it is possible not only to achieve neck pain relief, but to help prevent greater problems down the road. Let’s take a look at some neck pain solutions you can use at home with products designed to help achieve relief and improve the health of your neck into the future.
Neck pain can result from many activities. It is common in people older than 50 years, but now there are much younger people suffering from this dreaded condition. Neck pain can occur anywhere on the neck, from the bottom of the head to the top of the shoulders or spreading along the arms and upper back. Sleeping with the neck twisted, painting a ceiling, slouching, falling from a certain height, whiplash from a car accident, working with computers, and many more activities can lead to a cervical spine conditions. It’s no wonder so many people are looking for neck pain solutions.
Pillows
There are many types of pillows and many claims made by those who sell them. A good pillow for the neck will permit a good night’s sleep while maintaining a relaxed and natural posture regardless of position. One of the best ways to get a good pillow is to do a little research on what the different types of neck pillows are trying to achieve.
It is important to have a wide selection of cervical pillows, because everyone is built different and will have different needs. The best pillows maintain the natural alignment and this helps to relax muscles and reduce pressure on the joints and discs. The therapeutic pillow has been proven over the years to be one of the best designed pillows for neck pain relief and is a functional pillow.
It uses separate areas for back and side sleepers and, by taking a simple measure, you can get the right pillow that will maintain support for the neck and upper back when back sleeping and keep this relaxed posture for the neck and shoulders when side sleeping. In most cases, a neutral spinal posture reduces strain and further insult to sensitive or inflamed tissues. Sometimes a specific pillow strategy is needed based on symptom response.
Too much rotation or later flexion side bending can cause a stiff neck, irritate joints and stress discs. This leads to pain and headaches. Pillows that incorporate separate areas for different sleeping positions require conscious positioning when changing sleeping postures. For those who do not feel they can change positions in a conscious manner, there are many choices of pillows that have a uniform surface.
Traction For Neck Pain Solutions
There are many different products and range of prices for neck traction devices. This allows for cervical spine traction that is safe, comfortable, and user control. Traction is designed to produce decompression of the spine by separating the bones and helping to relieve pressure on joints and discs.
It helps to relieve the pressure of gravity from the neck having to support the weight of the head. It also helps to relax muscles and can be used to rehabilitate injuries. Often used for herniated discs and arthritis, there are devices that allow you to control the exact amount of force to obtain your own personal level of relief.
There are many popular traction products for home use as technological advances for neck pain solutions progresses. Traction can be also achieved by the use of special pillows that allow just the weight of the head and an ergonomic design to provide mild levels of traction. This can often help with restoring the natural curve of the neck. Other models are popular because they fit like collars and can be used when sitting. Some use these to help prevent the neck from being bent too far down when working. There are inflatable collar units that use separate left and right controls which can help with certain types of cervical spine disc problems, scoliosis and with rehabilitation of neck injuries.
The home neck traction helps bring nutrition into the disc, while restoring the natural curve. The type depends of the level of traction needed to achieve neck pain relief and the desired effect regarding long term goals for restoring biomechanics and treating hypolordosis or “military neck”.
Most types of traction provide linear or straight traction with varying levels of force, some quite strong. Some, like in the video above produce a pumping action with mild levels of force. For restoring the curve the pumping action units and those that place the cervical spine in extension, like some of the pillow types, are usually the best. For those who are treating disc herniations, bulges, and protrusions, pinched nerves, and degenerative arthritis, the straight line, higher force units will offer the best relief.
Collars
The use of collars for the neck may be questioned by some health care professionals. In general, they range from those that completely immobilize to those that provide very little support, if any.
Neck collars may help to provide pain relief in circumstances when there is injury to the cervical spine. When traction is not indicated because of inflammation, softer collars can help provide relief by providing a measure of support to help relieve muscle spasm, providing protection from temperature extremes and permit a measure of emotional security.
While there are not a great amount of designs to choose from; the simple foam ones are ubiquitous, there are those that provide a better design or construction that provide actual benefits other than to remind one of better posture or not to move in directions that hurt.
While some like to sleep with collars, this is not recommended and can distort the purpose of an ergonomic pillow. However, many cheap braces do essentially nothing, therefore; no harm will be done using them for extended periods or sleeping. It can help to provide a measure of warmth and emotional support. However, a brace that is well designed and uses quality materials can actually provide benefits for neck conditions. These should be worn with an intended balance between restriction of motion and levels of beneficial activity.
Exercises
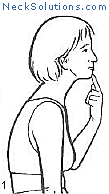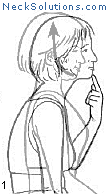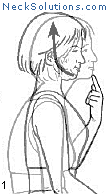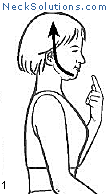 Exercising the cervical spine is an important part of achieving neck pain relief as well as rehabilitation and prevention. As opposed to other methods which are generally passive, active participation in improving neck muscle function is a necessary part of any system to improve function as well as to relieve pain.
Exercising the cervical spine is an important part of achieving neck pain relief as well as rehabilitation and prevention. As opposed to other methods which are generally passive, active participation in improving neck muscle function is a necessary part of any system to improve function as well as to relieve pain.
Exercises do not have to be done using any particular product, however, some of these products are very good and provide a means of motivation that can help you comply with a regular routine and provide more advanced methods.
Some of the exercises are very simple, yet effective. The exercise that consists of bringing your head over the shoulders and then performing a nodding motion like you are motioning “yes”, helps to strengthen the deep muscles of the neck that are responsible for maintain the natural curve. When this is mastered, a stretching of the neck with rotating to either side can be done, and this helps to maintain range of motion and flexibility. This is a great place to start and costs nothing.
Just doing the nodding motion has been shown to be one of the best methods to relieve neck pain and headaches. The neat thing about this is it can be done anywhere, even sitting at a computer or driving to provide therapy and relief. Some will need to progress to strengthening the neck and shoulder muscles in order to obtain long term relief. There are many exercises to choose from and products to help.
Using exercise for relief cannot be stressed enough. As stated, it does not have to be an extensive or complicated program, simple motions can be very effective. But it is the least liked method. We tend to just want to put something on or lay on something that will help. Exercise requires active participation, but we can’t get lazy if we truly want to effect not only symptoms, but long term success.
Travel Pillows
Travel pillows are not only for traveling or on the road, but can be used at home to help when relaxing or reclining to help maintain comfort, support and alleviate postural strain.
There are special versions of larger pillows to make using a good pillow easier for packing. The most popular of these pillows are the u-shaped pillows. They make traveling in a car or just about any vehicle more neck friendly. They can also be used lying down. They get specialized in shape and form for extra support
Remember, holding the head in awkward postures for long periods is very detrimental. It is a form of progressive minor injuries resulting in long term consequences. This is like repetitive strain injuries, where frequent, repeated wrist extension or flexion results in elbow pain called later or medial epicondylitis (tennis or golfers elbow), or carpal tunnel syndrome.
So, just like ergonomic interventions can be a key to long term control of these issues beyond initial therapy, a special pillow designed for sitting that keeps your head in better posture is essential regardless of if it is on the road or at home.
Heating & Cold Therapy
Heat is to be used carefully. If you have an injury, heat should not be used for the first few days when there is swelling and inflammation. Once this has calmed down, heat is a great way to relax muscles and increase blood flow for further healing and neck pain relief.
Cold therapy is recommended for initial stages of injury to reduce inflammation and further injury. There are conditions where it is questionable and you should go with what works. This does not mean necessarily what feels good; heat can feel good on a recent injury, but will be causing further damage and prolonging healing.
Specially designed heating pads deliver moist heat and deep penetrating infrared heat to specific areas of the neck and shoulders. Using heat therapy for the neck has never been easier or more effective. While you do not need any product to deliver heat, a hot shower is good, these products make it easier to apply on a more consistent basis, therefore, making it more effective to achieve neck pain relief just about anywhere.
Special wraps are a perfect way to provide cold therapy. Many can be used for both heat and cold. This is very economical and beneficial. After the initial acute stage of an injury; for the first few days, there is a period where alternating hot and cold becomes beneficial, prior to switching to heat alone. With the advent of technology, these products are cost effective as well and should be handy for easy use of neck pain solutions.
Muscle Therapy
Muscle therapy for the neck can be passive, as massage rollers help to knead muscles as well as provide passive motion therapy to the joints. Other massagers require you to be active in treating specific muscles or muscle groups.
The neck massagers are commonly seen in many places. While most are meant to be used on other areas like the lower back and legs, simple ones are meant to be used only on the neck as the rollers are closer together for the smaller area of the neck. The massage it produces is not overpowering, it is relaxing and also meant to provide some motion into the neck which can help with bringing in nutrition to the discs and joints of the cervical spine.
Other models are meant to treat specific areas of muscle strain or areas of trigger points, which can cause local pain as well as refer pain to other areas like the head and arms. These massage tools can help treat deep into the muscle, releasing areas of stiffness and trigger points. Being active in this process helps to more effectively treat your own neck, whenever needed. There are products that you lie down on and move your neck using gravity and the tool, and those that are ergonomically design to use leverage advantage for increased pressure. These methods require some time to practice and can be a little painful, consistent with deep muscle therapy.
Topicals & Supplements
There are many benefits to the use of topical pain relievers and supplements. For those who do not like cold on the neck, some topicals can produce the effects of cold therapy and pain relief as well. They are generally well tolerated. Some use ingredients to mask the pain as well as to help reduce inflammation and heal.
Supplements are helpful, especially when there is a deficiency of a particular vitamin. Vitamin E is one of these and has been shown to help with reducing pain and inflammation associated with degenerative conditions. Similarly, supplements of vital joint nutrients can help ease the pain of arthritis, like glucosamine and hyaluronic acid. New information is being discovered and we keep up with this to sort out the maze of joint health supplements.
There are many proven benefits of many supplements known to reduce inflammation. The combination of topicals and supplements can increase the benefits of both. There can be a synergistic effect using both together, and we also get into some herbal formulas as well. TENS therapy is also a topical means to reduce or interfere with nerve signals that produce pain.
Neck Pain Solutions – More
It is not always easy to achieve your goals for relieving pain. When neck pain becomes chronic, it can be a difficult and frustrating road to recovery. That is why the sooner you seek neck pain solutions, the better the outcome. There can be some symptoms associated with the neck that are normally not considered to be related, and these neck pain symptoms can be disturbing like dizziness and nausea. Emotional problems often need to be tackled with long standing neck pain and disability.
There are some factors that increase the risks of a neck pain. This is an important factor in seeking neck pain solutions. Age is one such factor as people over 50 years can more likely have a degeneration of discs, joints and bone spurs in the vertebrae of the neck. Recent injury to the neck and adjoining regions can be another risk factor. Rheumatoid Arthritis, which can affect the soft tissues and bone structure of the neck, is another risk factor. Being aware of these risk factors could help to minimize its effects on your neck pain.
Hopefully, these simple neck pain solutions can help prevent long term problems and be a positive factor in managing neck pain. Get help when you need it. There are instances when neck pain is a medical emergency. However, most neck problems are not serious and using these methods has helped many individuals with achieving neck pain relief. It is also important to learn proper ergonomics at home and at work, to learn safe lifting methods, which are not just for the back, and help managing longstanding problems like whiplash.
A 2020 study in Musculoskeletal Science & Practice found that those experiencing neck pain displayed slower hand and foot reaction and response times as well as impaired hand-eye coordination, indicating deficits in sensory and motor functions.
A 2021 study in BioMed Research International found neck pain is associated with incorrect head posture, limited range of motion in the cervical spine and TMJ dysfunction. The data suggests therapy focusing only on the neck region may improve neck pain and condition of the TMJ.
A 2022 study in BMC Medical Education indicates patient centered care is recommended in clinical practice to improve outcomes and reduce health care costs. However, the results of the study indicate health care students have low attitudes towards patient centered care. The authors recommend studies to evaluate if attitudes towards patient centered care can be improved during education.
It is important to find a balance between treating the patient and the changing system towards time restrictions and lumping patients into categories to facilitate minimizing time and maximizing profit. Often, providing patient centered care is difficult when decisions are made by insurance companies and administrators. In my experience, there is a distinct advantage for the patient both in terms of treatment response and psychological considerations when the patient knows you are sincere in caring for them personally.

 Neck Traction Devices
Neck Traction Devices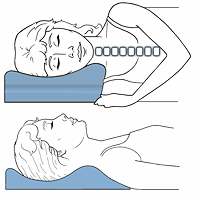 Cervical Pillows
Cervical Pillows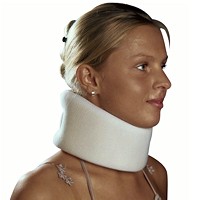 Neck Support Collars
Neck Support Collars Muscle Therapy Tools
Muscle Therapy Tools Head Supports
Head Supports Topical Pain Relievers
Topical Pain Relievers Special Pillows
Special Pillows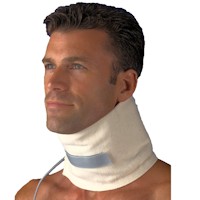 Heat Therapy
Heat Therapy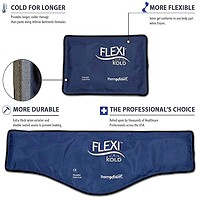 Cold Therapy
Cold Therapy TENS Therapy
TENS Therapy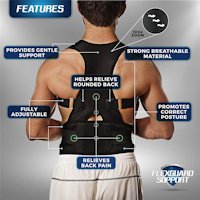 Posture Braces
Posture Braces Neck Stabilization
Neck Stabilization Ergonomic Aids
Ergonomic Aids New Mattresses
New Mattresses Relief Supplements
Relief Supplements